Concentrated on mobile post-transcription mechanisms within the CD73 ectoenzyme would possibly advertise anti-tumor immunity and sluggish most cancers development in triple-negative breast most cancers, in line with a Northwestern Medication find out about printed in Science Advances.
The find out about, co-led by way of Bin Zhang, MD, Ph.D., professor of Medication within the Department of Hematology and Oncology and of Microbiology-Immunology, suggests a brand new immunotherapy technique for sufferers who recently lack efficient remedy choices.
“For triple-negative breast most cancers, you wish to have to imagine concentrated on a big immunosuppressive mechanism, and concentrated on CD73 has now grow to be an rising possibility along with different standard checkpoint blockades,” mentioned Zhang, who could also be co-leader of the Tumor, Surroundings & Metastasis (TEAM) Program on the Robert H. Lurie Complete Most cancers Middle of Northwestern College.
Triple-negative breast most cancers (TNBC) cells don’t include the standard hormone and protein receptors usually present in breast most cancers cells, which leaves a restricted collection of viable healing goals.
Along with surgical operation, radiation and chemotherapy, immunotherapies similar to immune checkpoint inhibitors—medication that establish and block explicit proteins or “checkpoints” produced by way of immune cells and most cancers cells—had been broadly used to regard forged tumors, together with TNBC. Alternatively, earlier medical trials have proven that the majority sufferers with TNBC have little to no reaction to that more or less treatment.
Within the present find out about, the investigators aimed to spot new healing goals that may mobilize the frame’s immune machine to triumph over tumor-induced immunosuppression from TNBC cells.
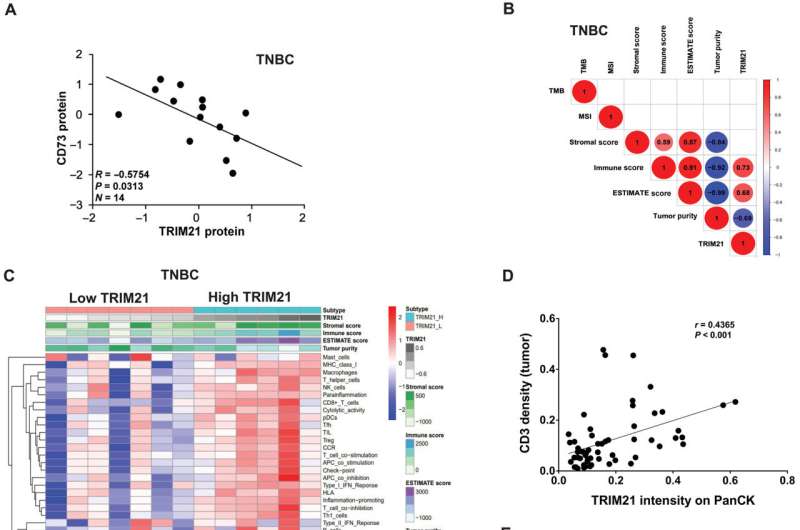
Via examining TNBC mobile strains, the workforce found out that increased ranges of the energetic ectoenzyme CD73 had been expressed at the floor of most cancers cells. This larger expression of CD73 is ordinary, in line with Zhang, suggesting that increased ranges of the enzyme building up immunosuppressive task inside the tumor microenvironment.
The use of complicated microscopy ways to analyze the most cancers cells additional, the investigators discovered that the ubiquitinase protein TRIM21 mediates the degradation of CD73, and disrupting TRIM21 stabilized CD73 and, in flip, suppressed CD8-positive T-cells that will have in a different way promoted an adaptive immune reaction in opposition to the most cancers.
“Subsequently, you’ll be able to in fact supply further choices to generate reagents to dam the structural interplay between those two molecules,” Zhang mentioned.
The investigators additionally extracted explicit amino acids from CD73, which degraded crucial intracellular purposes of CD73, particularly ubiquitylation, and enhanced tumor expansion by way of fighting antitumor immunity.
General, the findings expose a brand new attainable healing technique by which mitigating CD73 protein ranges may just save you TNBC tumor development.
Lowered ranges of CD73 and larger ranges of TRIM21 in most cancers cells may just additionally function biomarkers for figuring out sufferers who could have a extra favorable reaction to immunotherapy, in line with Zhang.
“We expect in the event you modulate CD73 protein ranges at once, now not best are you able to diminish the enzyme task but in addition can goal CD73 impartial of enzyme task serve as,” Zhang mentioned.
Additional information:
Ziyi Fu et al, Proteolytic law of CD73 by way of TRIM21 orchestrates tumor immunogenicity, Science Advances (2023). DOI: 10.1126/sciadv.add6626. www.science.org/doi/10.1126/sciadv.add6626
Quotation:
Novel healing goals found out for triple-negative breast most cancers (2023, January 19)
retrieved 22 January 2023
from https://medicalxpress.com/information/2023-01-therapeutic-triple-negative-breast-cancer.html
This record is topic to copyright. Excluding any truthful dealing for the aim of personal find out about or analysis, no
section could also be reproduced with out the written permission. The content material is supplied for info functions best.
Supply Via https://medicalxpress.com/information/2023-01-therapeutic-triple-negative-breast-cancer.html